Tuberculosis has been a scourge of humanity for millennia: the oldest known cases are in a pair of 9,000-year-old skeletons. Despite modern therapies, the disease affects about ten million people a year. It’s challenging both to treat, and to study in the laboratory.
Part of the difficulty is that the bacterium that causes it, Mycobacterium tuberculosis, holes up inside the very immune cells that the body deploys to fight it. Any treatments must therefore cross the human cell membrane before even attempting to attack the bacterium — a tricky manoeuvre. Similarly, researchers studying host–pathogen interactions must find ways to study the bacterium in cultured cells or in animals to capture the true complexity of the infection.
And it’s not just M. tuberculosis. Many other intracellular pathogens, including Salmonella typhimurium, Chlamydia trachomatis and Listeria monocytogenes, are best investigated in tandem with their host cells. “We basically pay as much attention to the pathogen as we do to the host,” says David Russell, an infection biologist at Cornell University in Ithaca, New York. “I really think you can’t interpret one without the other.”
In the past, scientists tended to consider infected cells in bulk, because they lacked the tools to investigate them individually. Now, Russell and others are combining methods to see how host-cell responses to pathogens, and pathogen responses to host cells, differ from cell to cell. Understanding infection at that level could lead to new vaccines or treatments, and add detail and nuance to conclusions drawn from bulk-cell analyses. “The field’s going to learn so much,” says Anna Coussens, an immunologist at the Walter and Eliza Hall Institute of Medical Research in Parkville, Australia.
Dual sequencing
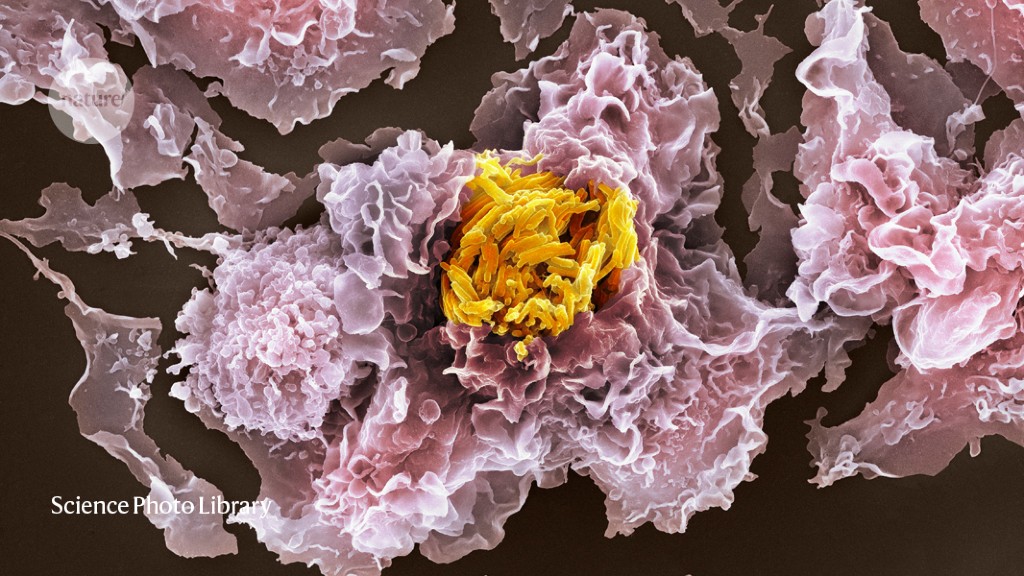
In the lungs, M. tuberculosis can infect two kinds of immune cell: alveolar macrophages that are resident there, and interstitial macrophages that are recruited from the bloodstream. Russell and his colleagues wanted to understand how these different cell types deal with the bacterium, and how it would respond in turn. To work that out, they decided to collect RNA sequences both from individual host cells and from the bacterium in live animals. No one had done this kind of sequencing from in vivo infections before, Russell says.
First, the team required a way to indicate how the microbes were faring in host cells. Specifically, the researchers needed a gene that the bacteria would turn on when they were struggling to survive. After years of testing, they selected a regulatory element from the stress-response gene hspx. They linked that to a gene for green fluorescent protein so that stressed pathogens would glow green. A second, always-on, red fluorescent protein served as a control.
The researchers infected mice with this two-colour reporter strain. After three weeks, they homogenized the lungs and sorted the cells into those that housed red, healthy bacteria and those that hosted red-green, sickly ones1.
The next step was to isolate and sequence the RNA of the host cells and the pathogens. This was far from trivial, says Russell. To avoid contaminating the host RNA with bacterial material, and vice versa, they had to isolate the RNA in a stepwise fashion. First, they used a chemical solution called Trizol to break open the host cells. Once they’d isolated the eukaryotic RNA, they used beads to break open the M. tuberculosis and extract its RNA.
Stress patterns
The resulting sequences revealed clear patterns in how the host and the pathogen responded to each other. Macrophages that mounted effective defences, indicated by the presence of RNA sequences for defensive genes, held more stressed-out bacteria than did those that failed to control the pathogens. Russell was surprised that the host and pathogen transcriptomes lined up so well.
Overall, interstitial macrophages tended to be more successful at controlling the bacterial infection than were alveolar macrophages; for example, they produced a protein that makes nitric oxide to attack the invaders. However, a subset of alveolar macrophages also responded in this way.
This kind of information could be useful to vaccine developers, says Russell. Although there is a vaccine for tuberculosis, it’s not effective in adults. Vaccine designers might overcome this, he suggests, by augmenting their formulations to promote an effective macrophage response.
The study also provides a long list of host genes that should point researchers to pathways that either promote or hinder bacterial survival.
Dual imaging
Sander van Kasteren, a chemical biologist at the University of Leiden in the Netherlands, calls the Russell study a tour de force. “That level of detail is just amazing,” he says.
He should know. Researchers in his lab have also been studying M. tuberculosis and Salmonella enterica at the level of single host cells, although in culture rather than in vivo2,3. They use a dual-imaging approach called correlative light electron microscopy (CLEM): it starts with fluorescence microscopy to view specific fluorescent markers and then uses electron microscopy to visualize the subcellular structure of the host cells.
To attach the fluorescent markers to the relevant molecules, the team used a technique called click chemistry. The classic click reaction joins a nitrogen-rich azide group with a triple-bonded carbon group called an alkyne in the presence of copper. The researchers fed M. tuberculosis an azide-containing analogue of the amino acid methionine, and an alkyne-containing version of the amino acid alanine that is used only in bacterial cell walls2.
After infection and fixation of the host cells, they added a green fluorophore containing an alkyne, which clicked on to the methionine analogue to label bacterial proteins, and a red fluorophore with an azide to visualize the bacterial cell walls. The resulting images, collected with confocal fluorescence microscopy, revealed roughly where in the cells the bacteria were located, and whether they’d shed their defensive cell walls. Usually, that would be the end of the microscopy experiment. But for lab member Thomas Bakkum, it was only step one.
Bakkum took the same slides and used transmission electron microscopy to visualize the cells at the nanoscale level. By overlaying the light and electron images in huge pictures — they would measure 144 square metres if he were to print them out — Bakkum could see the shape of the bacteria and any cellular organelles they associated with in fine detail. Dead microbes, he found, tended to accumulate in large cellular compartments called vacuoles, whereas live ones were generally found in the cytosol, the substance filling the interior of the cell.
The most logical use for this technique, Bakkum reckoned, would be to test new antibiotics in cultured cells. CLEM would allow researchers to pinpoint how the drugs affect where bacteria accumulate and whether they lose their cell walls. As a proof of principle, he tested a few tuberculosis antibiotics on his cultures.
Like Russell, Bakkum and his colleagues saw that bacterial cells do not always react in the same way to seemingly uniform cellular conditions. Even within a cell, some bacteria were destroyed; some didn’t change; others adopted a bizarre, angular shape, says van Kasteren. Some of these diverse responses to treatment might help bacteria to survive, he speculates.
Drug reservoirs
Maximiliano Gutierrez, a cell biologist at the Francis Crick Institute in London, has also used CLEM to study antibiotic efficacy, but with an analytical-chemistry twist. For a study published in 2019, Gutierrez wanted to localize the bromine atoms in the antibiotic bedaquiline, used to treat drug-resistant tuberculosis, to see where the drug goes. After light microscopy and electron microscopy, his team subjected the samples to a form of mass spectrometry called ion microscopy, which collects molecular weight and localization data4.
The analysis revealed that bedaquiline tended to accumulate in lipid droplets in the host cell. The droplets served as a sort of reservoir for the antibiotics, because the bacteria would mine them for energy, and pick up the drug in the process. Although pharmaceutical companies usually try to avoid fat-soluble drugs because they don’t last long in the body, Gutierrez says his study suggests that they shouldn’t be so hesitant to consider using such compounds to treat tuberculosis.
Gutierrez says that the triple-imaging approach was crucial to their discovery. It is “the only way you can visualize where the drugs go”, he says. And these types of multimodal studies could be the future of host–pathogen research, adds van Kasteren. But, he says, researchers could go even further — perhaps combining his click-chemistry tagging approach to identify cells of interest with Russell’s dual-sequencing technique to home in on the responses. After some 9,000 years, such combinations could finally give scientists a powerful advantage in the quest to control the disease.





More News
Author Correction: Stepwise activation of a metabotropic glutamate receptor – Nature
Changing rainforest to plantations shifts tropical food webs
Streamlined skull helps foxes take a nosedive